Any failure of the medical device and the precise description of its cause must be reported to the person in charge of the safety of medical products
at
daum electronic gmbh
.
The device must be tested according to GEF (German Electrotechnology Federation) 0751-1 before it is put back into service.
Failure description
Spare part / Item number
Tester
Signature
Date
19
Service Case
Flugplatzstr. 100
D-90768 Fürth
Tel. ++49 (0) 911/9 75 36-0
Fax ++49 (0) 911/9 75 36-96
GB
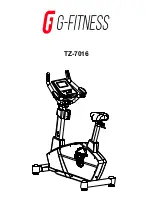
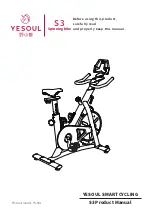
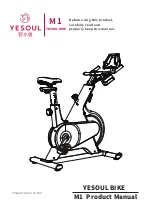
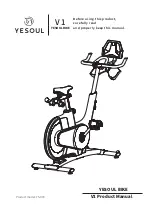
Summary of Contents for ergo_bike medical8
Page 1: ...medical Operating Instructions 0650682e 0004 0297...
Page 23: ......