6
.
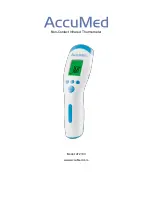
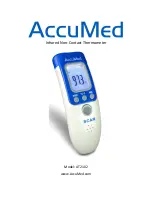
Description of Symbols
0197
The device is in accordance with Medical Device
Directive 93/42/ EEC
FDA Approved
(2)
1.5v A
A
A Batteries
Type B equipment
In order to protect the environment, please recycle the
battery according to the local regulations
Body
Object
Indication of Mode
Attention! Consult accompanying documents.
XLE-MD-TM-IR-R1
Larson Electronics, LLC
P
:
(800) 369-6671 www.larsonelectronics.com
5